- Blog
- Chrome extensions website blocker
- Ejemplo de intervalo variable
- Mini vmac ios 10 no jailbreak
- Dopewars for android
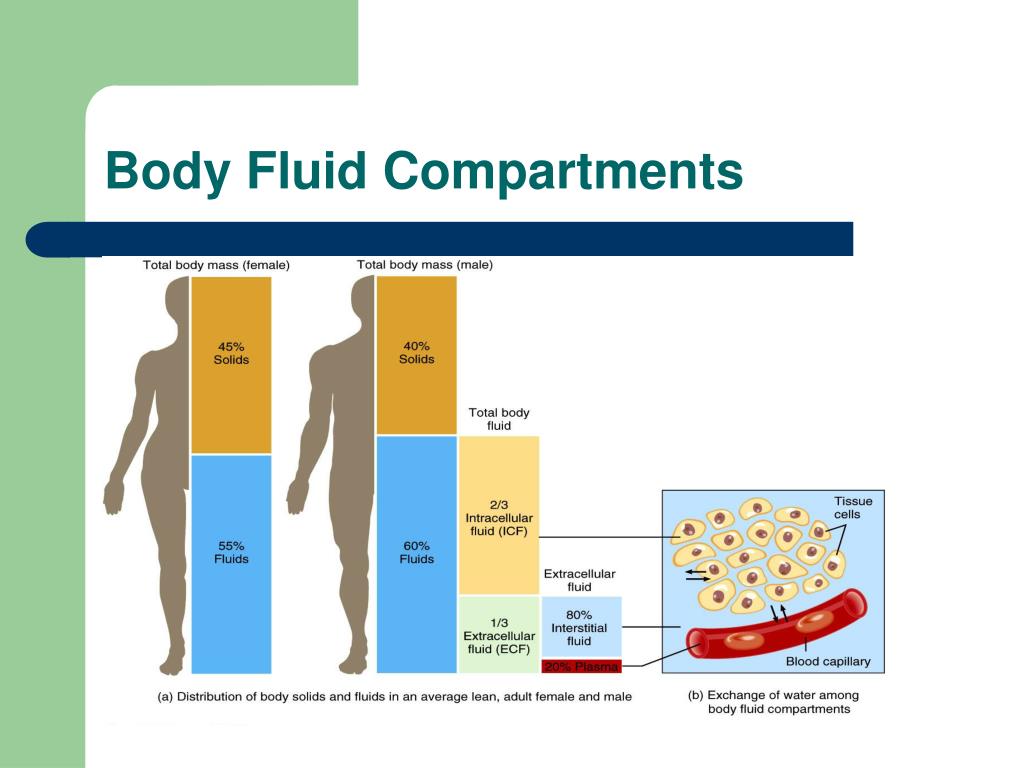
- Electrolyte composition of body fluid compartments
- Taurus g3 9mm msrp
- Gimp for mac os x lion download
- Free ram optimizer windows 7
- Master pdf editor 4 free download
- Nike run swift wide
ECW is under neuroendocrine control, and the final regulation is accomplished by the kidney through retaining or excreting solutes and fluids. The volume and composition of body fluid compartments are strictly controlled. It is helpful to keep this classification of the processes underlying accumulation of uremic toxins in mind when approaching a patient with the uremic syndrome.Įndre Sulyok MD, PhD, DSc, in Nephrology and Fluid/Electrolyte Physiology: Neonatology Questions and Controversies (Second Edition), 2012 Cell Volume Regulation PTH excess in CKD is a prime example of a Type II uremic toxicity. As outlined earlier, dysbiosis of the intestinal microbiota is a very important contributor to the pathobiology of uremia, and is embraced in the Type I mechanism. These categories of pathobiological process are not mutually exclusive, and one may predominate over another in certain circumstances. For example, urea is a putative uremic toxin that arises because of a combination of Type I and Type III processes-excessive accumulation due to impaired renal excretion and continued production due to exogenous (dietary) consumption of protein as a precursor of urea. Combinations of more than one pathobiological process are possible. A Type IV mechanism is a deficiency or reduced activity of substances normally produced endogenously as a result of decreased synthesis, enhanced degradation, or biological inhibition. A Type III mechanism is the accumulation of toxic substances in biological fluids from exogenous sources by virtue of reduced renal excretory capacity often combined with continued usual dietary consumption.

A Type I mechanism represents the accumulation in body fluids of toxic substances normally produced endogenously by metabolic processes, including those that occur in intestinal microbiota, largely as a result of reduced renal excretory capacity (decreased GFR and/or tubular excretory function) A Type II mechanism is a surfeit of toxic substances in body fluids as a result of excess endogenous production or impaired degradation (or both) but not necessarily directly due to reduced renal excretory capacity. Uremic toxins can accumulate in body-fluid compartments through a number of distinct mechanisms. Massry, in Nutritional Management of Renal Disease (Fourth Edition), 2022 The pathobiologic categorization of uremic toxins according to the processes underlying accumulation in body fluids The bound water fraction appears to be related to the osmotically inactive body sodium mainly stored in glycosaminoglycan-rich tissues (Titze et al, 2003), which provides a buffer system in the control of physiologic dehydration. Water can be liberated from this latter bound fraction in a regulated manner irrespective of its location in the cellular or extracellular space.
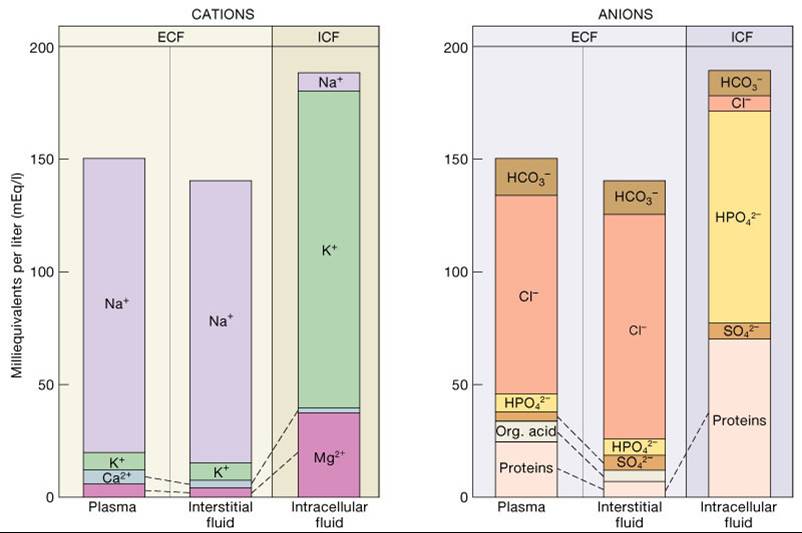
#Electrolyte composition of body fluid compartments free#
Two distinct water fractions should be considered: the free “bulky” water and the relatively slow-motion bound water. The term physical water compartments designates the physical states of tissue water and assumes interactions between dipole water molecules and tissue biopolymers, including proteins and glycosaminoglycans.

The physical compartmentalization and mobility of tissue water could play a significant role in neonatal body fluid redistribution. According to a recent hypothesis, cell water rather than extracellular water may be the source of significant neonatal water losses (Sulyok, 2008). The early postnatal weight loss observed in the first days of life is usually ascribed to the isotonic contraction of ECW volume and the elimination of excess sodium and water by the kidney. Redistribution of body fluid compartments occurs soon after birth. Jean-Pierre Guignard, Endre Sulyok, in Avery's Diseases of the Newborn (Ninth Edition), 2012 Neonatal Redistribution of Body Water Compartments
